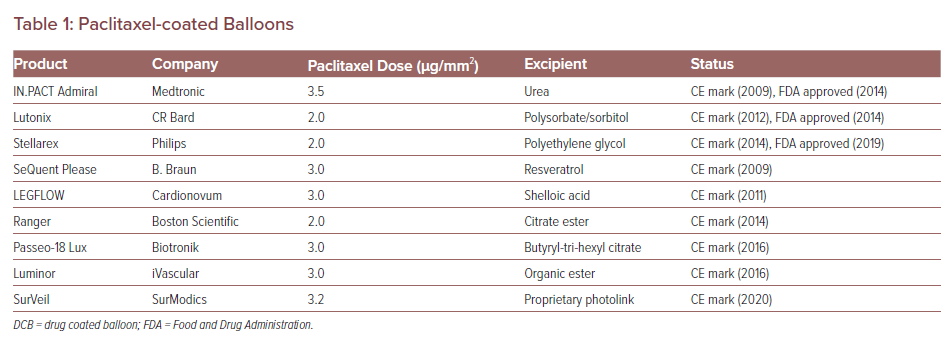
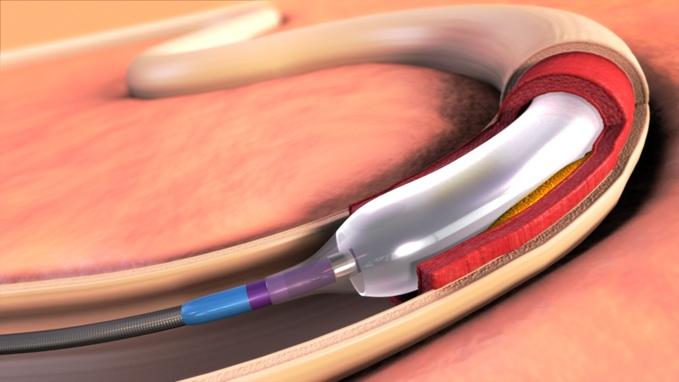
IN.PACT™ Admiral™ drug-coated balloon: Durable, consistent and safe treatment for femoropopliteal peripheral artery disease - ScienceDirect

Long-Term Clinical Effectiveness of a Drug-Coated Balloon for the Treatment of Femoropopliteal Lesions | Circulation: Cardiovascular Interventions

New MEDTRONIC In-Pact Admiral Paclitaxel-Coated PTA Balloon Catheter 4mm x 80mm x 130cm (X) ADM04008013P Disposables - General For Sale - DOTmed Listing #2847198:
Reimbursement summary for angioplasty of arteries of lower extremities | Med Tech Reimbursement Consulting
IN.PACT™ Admiral™ Instructions for Use Caution: Federal (USA) law restricts this product for sale by or on the order of a ph

Medtronic Issues Voluntary Recall for Subset of IN.PACT Admiral and IN.PACT AV Drug Coated Balloon | DAIC